- Home Page
- Company Profile
-
Our Products
- Aromatic Hydrocarbon Solvents
- De Aromatize Hydrocarbon Solvents
- Aliphatic Hydrocarbon Solvents
- Cyclo Hexane Solvent
- Drilling Oils - Fluids
- Mining Oil
- Superior Kerosene Oil
- Hexane Solvent
- Mineral Turpentine Oil -MTO
- Slop Oil
- Heptane Solvent
- Aluminium Rolling Oil
- Nitrobenzene Chemical Structure
- MIBK (Methyl Isobutyl Ketone)
- MEA (Monoethanolamine)
- MDC (Methylene Dichloride - Dichloromethane)
- Low Aromatic White Spirit (LAWS)
- LDO (Light Diesel Oils)
- LABSA (Linear Alkyl Benzene Sulphonic Acid)
- Iso Propyl Alcohol
- DMSO (Dimethyl Sulfoxide)
- Butyl Cellosolve (Ethylene Glycol Monobutyl Ether)
- Butyl Acetate
- Benzene Chemical Structure
- Octanol Chemical Structure
- Rubber Process Oil
- Glycols
- Petroleum Ether
- Base Oils
- Deodorised Kerosene (DOK)
- Organic Solvents
- Industrial Oil
- Contact Us
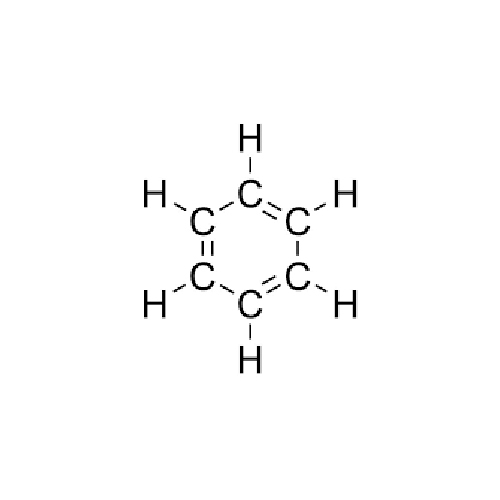
Benzene Chemical Structure
Product Details:
- Molecular Weight 78.11 g/mol
- Ph Level Neutral (not applicable)
- Odour Aromatic, sweet odor
- Solubility Insoluble in water; soluble in alcohol, ether, acetone, chloroform
- Density 0.8765 Gram per cubic centimeter(g/cm3)
- Appearance Colorless, transparent liquid
- Storage Instructions Store in a cool, dry, well-ventilated area away from heat and ignition sources
- Click to view more
X
Benzene Chemical Structure Price And Quantity
- 1 Drum
- Complies with REACH, OSHA, and related regulations
- Stable under normal temperatures and pressures
- Flammable Liquid (Class 3)
- 5.5C (41.9F)
- 497C (927F)
- 1.501
- C6H6
- Steel Drums, ISO Tanks
- 9.8 kPa at 20C
- 80.1C (176.2F)
- 11C (12F)
- Harmful, Carcinogenic
- 29022000
- 0.652 cP at 25C
Benzene Chemical Structure Product Specifications
- Cyclohexatriene, Benzol, Phenyl hydride
- Solvent (paints, varnishes, resins), Intermediate for chemicals, Laboratory reagent
- 78.11 g/mol
- Colorless, transparent liquid
- Industrial, Laboratory, Chemical synthesis
- Neutral (not applicable)
- Organic compound
- Petroleum, Coal tar
- Liquid
- 2 years from manufacturing date
- Aromatic, sweet odor
- Store in a cool, dry, well-ventilated area away from heat and ignition sources
- Stable under recommended storage conditions
- Other
- 99.9%
- Benzene
- 71-43-2
- 0.8765 Gram per cubic centimeter(g/cm3)
- Insoluble in water; soluble in alcohol, ether, acetone, chloroform
- Not applicable (volatile liquid)
- Complies with REACH, OSHA, and related regulations
- Stable under normal temperatures and pressures
- Flammable Liquid (Class 3)
- 5.5C (41.9F)
- 497C (927F)
- 1.501
- C6H6
- Steel Drums, ISO Tanks
- 9.8 kPa at 20C
- 80.1C (176.2F)
- 11C (12F)
- Harmful, Carcinogenic
- 29022000
- 0.652 cP at 25C
Benzene Chemical Structure Trade Information
- 5000 Drum Per Month
- 2-10 Days
Product Description
An aromatic hydrocarbon solvent used mainly as a raw material for chemical synthesis but restricted due to toxicity.
Applications:
- Chemical intermediate production
- Resin and polymer industry
- Laboratory solvent (with restrictions)
- Pharmaceutical synthesis (controlled use)
Chemical Structure and Physical Properties
Benzene features a unique ring structure (C6H6) with alternating double bonds, giving it aromatic stability. It has a melting point of 5.5C, a boiling point of 80.1C, and is a volatile liquid with a refractive index of 1.501. Benzenes distinctive sweet smell, colorless appearance, and low viscosity make it recognizable in laboratory and industrial settings.
Applications and Usage
Benzene serves as a versatile solvent in paints, varnishes, and resins, and acts as a crucial intermediate in the synthesis of chemicals such as plastics, dyes, and pharmaceuticals. It is also utilized in research labs as a reagent. Generally, benzene is directly used or blended with other chemicals, depending on the intended application.
Safety and Handling
Due to its flammable and carcinogenic nature, strict safety protocols must be followed when handling benzene. Store benzene in cool, dry, well-ventilated areas away from heat and ignition sources. Industrial users must comply with regulatory guidelines, including those set by REACH and OSHA, and use appropriate protective equipment during handling.
FAQs of Benzene Chemical Structure:
Q: How should benzene be safely stored and handled?
A: Benzene must be stored in tightly sealed steel drums or ISO tanks in a cool, dry, and well-ventilated area away from heat and ignition sources. Proper personal protective equipment should be worn due to its flammability and carcinogenic risk, and storage areas should comply with REACH, OSHA, and related regulations.Q: What are the main industrial uses of benzene?
A: Benzene is primarily used as a solvent in paints, varnishes, and resins. It also functions as an intermediate in the production of chemicals such as plastics, dyes, rubber, and pharmaceuticals, and is employed as a laboratory reagent for chemical reactions and analyses.Q: When is benzene typically used during chemical processes?
A: Benzene is typically introduced during the initial stages of chemical synthesis, where it acts either as a solvent or as a starting material in the creation of more complex organic compounds. Its chemical stability and solubility in organic solvents make it an essential component in various industrial workflows.Q: Where does industrial benzene usually originate?
A: Industrial benzene is mainly derived from petroleum and coal tar through refining and extraction processes. These sources are processed to yield high-purity benzene (99.9%), suitable for use as a solvent and intermediate in chemical manufacturing.Q: What benefits does benzene offer as a solvent or raw material?
A: Benzenes high volatility, ability to dissolve a wide range of compounds, and chemical stability under normal conditions make it highly effective as a solvent. As a raw material, its aromatic structure allows for efficient synthesis of many valuable chemicals in industry.Q: What are the regulatory and safety classifications associated with benzene?
A: Benzene is classified as a Flammable Liquid (Class 3), hazardous, and carcinogenic. It is regulated under international safety frameworks such as REACH and OSHA, requiring stringent handling, storage, and labeling protocols to ensure workplace safety and environmental protection.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email







 Call Me Free
Call Me Free
